Jaw bone defect therapy uses stem cells and fibrous scaffold combination. Ameloblastoma sufferers experience jaw bone defects after surgical therapy. The bone remodeling process takes a long time. However, several therapies have been developed to deal with this case.
Ameloblastoma is characterized by a slow-growing neoplasm cause invasive damage to a specific area or locality. Ameloblastoma generally occurs in 80% of the mandible with a preference for the posterior and maxillary regions. Mostly this type of case is often found in serious condition. The sufferers often find deformities on the face and due to malocclusion of teeth.
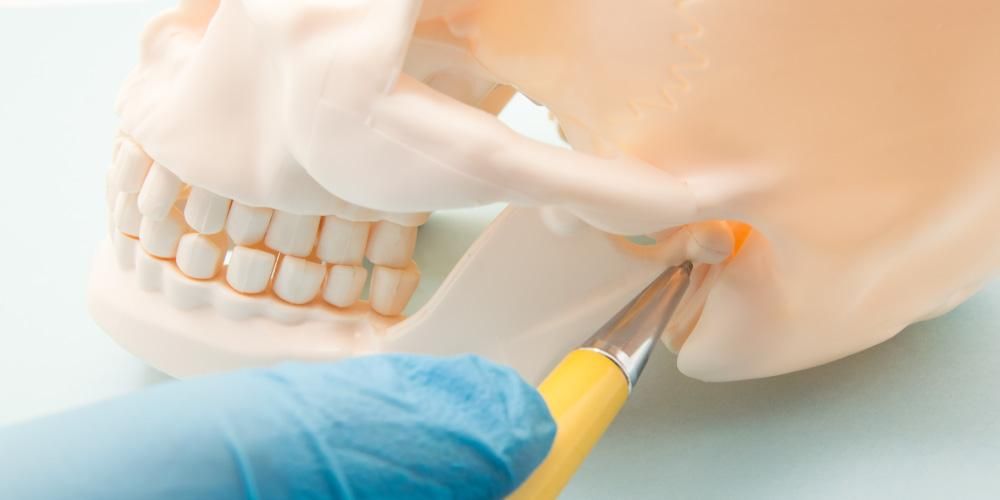
The bone defect can only be done regeneratively to return the bone structure to be normal. One of the regenerative treatments that can be done is by using stem cells. The use of dental stem cells has been developed recently. One of the uses of dental stem cells is obtained from dental follicle tissue. Dental follicles have a role in regulating bone remodelling by producing growth factors and cytokine markers (such as to regulate the formation of osteoblasts and osteoclasts during the eruption process). Besides that, the use of dental follicle stem cells has the advantage of being able to repair the cells themselves and to get them does not require invasive action and cause severe pain.
The use of dental follicle stem cells can be combined with the use of the scaffold, one of which is the fibrous scaffold nanocomposite. Nanocomposite fibrous scaffold is a scaffold structurally shaped similar to the original bone using a nanocomposite material derived from a nanohydroxyapatite matrix (65%) reinforced with 35% collagen fibrils and a fibrous structure obtained from silica material coated with nanohydroxyapatite and gelatin reinforced with electrospun PLLA thread %). Moreover, the combination of dental follicle stem cells and fibrous scaffold nanocomposites has the potential to regenerate postoperative bone defects in ameloblastoma.
The application of dental follicle stem cells (DFSC) and fibrous scaffold nanocomposites combination aims to make tissue DFSC regenerate bone defects faster. Fibrous scaffold nanocomposites contain nanoHA, which can be the material of choice for supporting osteoblast cell-bound and proliferation and mineralized bone matrix formation in vitro. Besides, studies have shown that chitosan is osteoconductive in vivo in surgery lead to bone breakdown. Other studies have shown that mixed fibrous scaffold nanocomposites work effectively at regenerating bone.
Some indicators, such as cell viability and mechanical strength, are excellent. Fibrous scaffold nanocomposite will undergo osteogenic differentiation which is played by gene expression of Runx2, ALP, Col, OCN and ALP activity. Runx2 induces osteogenic differentiation in the early stages and inhibits them in the late stages. ALP is essential in ECM mineralization with its expression usually indicative of the development of osteogenic differentiation. Col1 and OCN are constituents of ECM, and their expression is used as middle and late markers of osteogenic differentiation. ALP activity shows that the fibrous nanocomposite scaffolding is osteoinductive.
Author: Saka Winias
Details of this work can be viewed on: