UNAIR NEWS – Visual impairment or blindness is still a serious problem in Indonesia. The result of a study from 2014-2016 showed there were 6.4 million people from 15 provinces throughout Indonesia experiencing eye disorders. From 6.4 million people with eye disorders, 1.3 million are blind, while the remaining 5.1 million fall into the medium and severe categories.
Blindness can be caused by several factors, one of them is corneal ulcer. Corneal ulcer is an eye disorder that can interfere with the vision function.
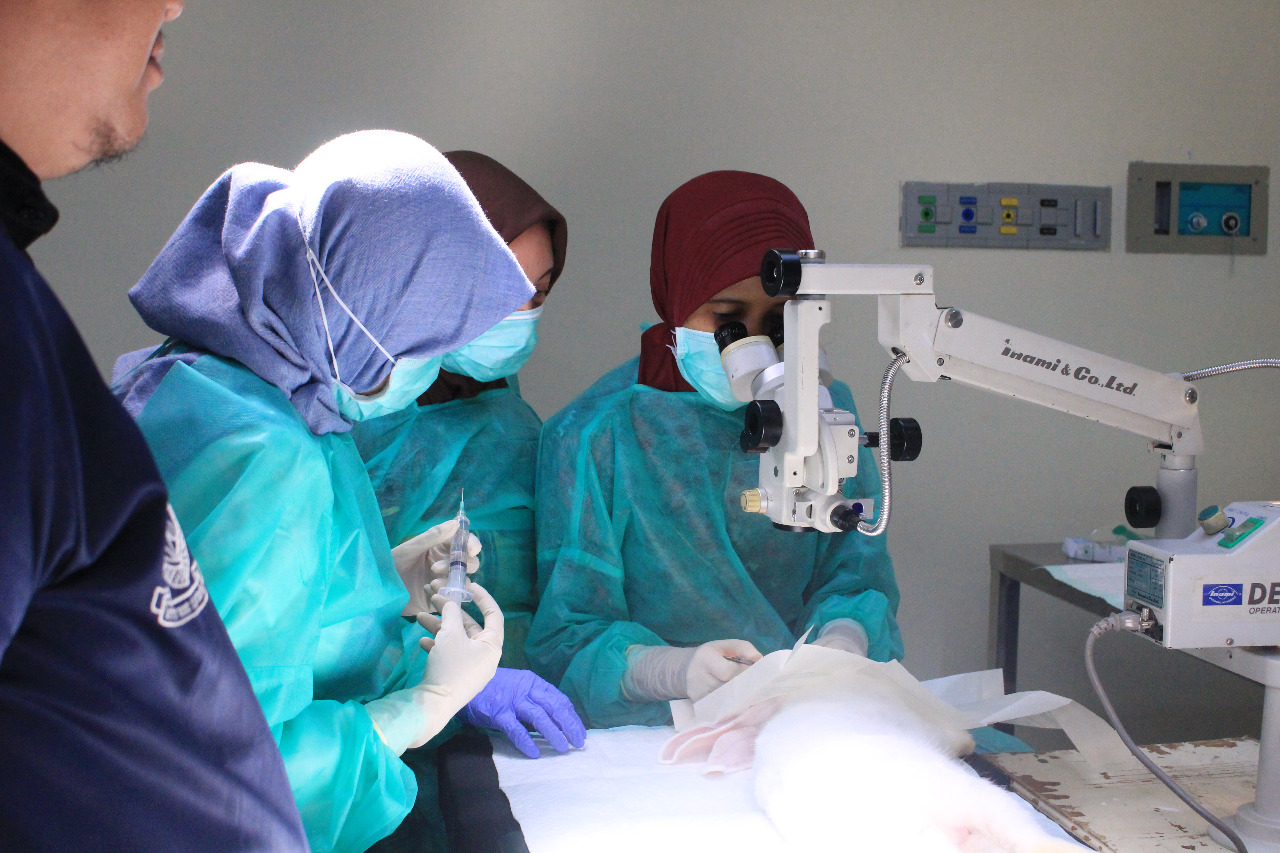
From this problem Dr. Prihartini Widiyanti drg., M. Kes, S. Bio, CCD, collaborating with ophthalmologists, and PPDS team (Participants of Medical Specialist Education Program) Ophthalmology Department Dr. Soetomo General Hospital – FK Unair created ” Collagen-Based Hydrogel as an Alternative to Blindness due to Corneal Ulcer”.
Dr. Yanti explained that the research that has been going on since 2015 continues to grow by varying the material to get the characteristics of artificial corneas to get meet to clinical needs, including its requirement to be moist in order to facilitate cell growth to support healing.
Material used
According to Dr. Yanti, in the development of corneal hydrogels there are several ingredients including collagen and chitosan. The collagen, she continued, is a type 1 collagen that is commonly found in the bodies of living things but mostly from cow collagen.
“There are several kinds of collagen sources such as red snapper scales, cow collagen and chicken feet. Type 1 collagen is found in all vertebrates and their characteristics are different, ” she explained.
While the chitosan used comes from the extraction of living things, shrimp and crab shells. Not only that, because the corneal hydrogel is synthetic, there is a mixture of chemicals.
“It depends on the characteristics we are aiming for, for example, if we emphasize in clarity, we will need synthetic materials, because natural materials have more impurities. But if we want to focus on the biocompatibility or the level of acceptance from the body, of course we use natural materials, ” she said.
Trials
The tests carried out, she continued, including in vitro tests and in vivo tests. According to Dr. Yanti, it passed many stages, ranging from chemical tests, physical tests and biological tests. Furthermore, in vivo or biological testing in living things is carried out together with a team consisting of ophthalmologists and PPDS team for insertion / implants to test animals.
“So there are stages, at the beginning of the test there is a lab scale test we call an in vitro test. It covers a lot starting from the confirmation of physical, mechanical, chemical and biological properties (cytotoxicity, which is testing whether the material is safe to be applied to the body) should we monitor first. Only then we will go to an in vivo test on living things, a biological test. So our research has reached the stage of implant in animals, assisted by an ophthalmologist. But we realize that it still requires a long step to be applied because it still requires optimization of some biomaterial characteristics, ” she explained.
With this research, Dr. Yanti hoped that in the future it will truly be a solution to the problem of injury due to ulcers or trauma to the cornea. (*)
Author: Asthesia Dhea Cantika
Editor : Binti Q. Masruroh